We are making efforts to shed light on the formation mechanisms of surfactant morphologies
6-3-1 Niijuku, Katsushika-ku, Tokyo 125-8585, JAPAN
RESEARCH
Hybrid Surfactants
A surfactant molecule having a fluorocarbon chain (CmF2m+1-) and a hydrocarbon chain (CnH2n+1-) in the molecule has been called "hybrid surfactant". We are investigating the synthesis and solution properties of hybrid surfactants, and have found their specific properties that have never seen for conventional surfactants such as hydrocarbon surfactants and fluorocarbon surfactants.
Viscoelastisity Behavior
When aqueous solutions of a hybrid surfactant is prepared, the solutions turn into a gel at 160 mM (10wt%) of surfactant. In the gel, there exsist a number of thread-like micelles which are entangling mutually. The following left picture shows the gelation of 10 mM of hybrid surfactant solution and right picture shows the exsistence of a number of thread-like micelles formed from the hybrid surfactant.

1) Y. Takahashi, Y. Nasu, K. Aramaki, Y. Kondo, J. Fluorine Chem., 145(1), 141-147 (2013).
2) Y. Takahashi, Y. Kondo, J. Schmidt, Y. Talmon, J. Phys. Chem. B, 114(42), 13319-13325 (2010).
Synthesis and Solution Properies of Stimuli-responsive Surfactants
Our group has so far synthesized stimuli-responsive surfactants and investigated on the solution properties of the surfactant. Now we are focusing on redox-active and photo-responsive surfactants.
Control over Solution Viscosity Using Photo-responsive Surfactants
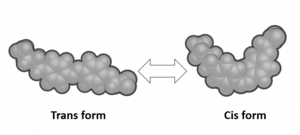
Our interests are surfactants having stimuli-responsive groups. We have synthesized photo-responsive surfactants having an azobenzene group. Azobenzenes can be isomerized from the trans isomer to the cis one under UV light irradiation and converted from the cis isomer to the trans one using visible light or heat.
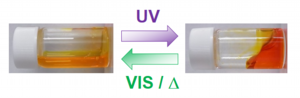
When UV light is irradiated to the aquous solutions of azobenzene-containing surfactants, the solution increases its viscosity because the surfactant molecules form thread-like micelles. On the other hand, when you irradiate a visible light to the solution, the solution viscosity is decreased. You can control over solution viscosities using light.
1) Y. Takahashi, Y. Yamamoto, S. Hata, Y. Kondo, J. Colloid Interface Sci., 470, 370-374 (2013).
Active Demulsification Using Photo-responsive Surfactants
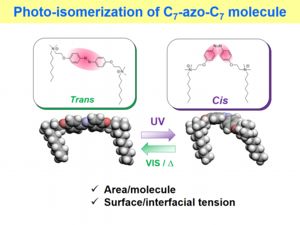
Using a gemini surfactant (C7-azo-C7) having an azobenzene group, you can accelerate the demulsification of stable emulsions consisting of water, octane, and the C7-azo-C7 using UV light irradiation. This is due to the change in the molecular area of C7-azo-C7 adsorbing at the interfaces between the water and octane phases in the emulsions.
1) Y. Takahashi, K. Fukuyasu, T. Horiuchi, Y. Kondo, P. Stroeve, Langmuir, 30(1), 41-47 (2014).
UV light was irradiated to the left bottle containing a emulsion. These emulsions were prepared by homogenizing a mixture of octane, water and a novel gemini surfactant having an azobenzene group. UV light irradiation brought about photoisomerization of the azobenzene group in the surfactant, followed by de-emulsification of the stable emulsion.
Finding of Metal-lustrous Organic Crystals
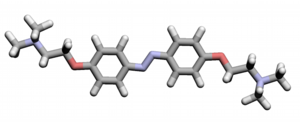
It is no exaggeration to say that the pursuit of technolo- gies to create noble metals, such as gold and silver, from base metals-that is, alchemy-has greatly contributed to the development of modern chemistry. In particular, gold has long fascinated people since ancient times because of its worth and beauty. In fact, gold-colored origami is the most popular form of origami among Japanese children who practice creating origami figures by folding colored papers available in commercial origami sets. We have found that azobenzene compounds can form gold-lustrous organic crystals. Metallic paints which contain metal fine particles have been used to obtain metal-lustrous films on substrates. Therefore metal-lustrous films obtained from conventional metallic paints are not transparent to radio waves. Our metal-lustrous organic crystals are electrically insulate, thus they can be used to colorize communication devices.
The above-shown compound gives gold-lustrous crystals (please see below).

In the crystals, the molecules are forming J-aggregates. Recently we have succeeded in preparing silver-lustrous crystals from organic compounds.
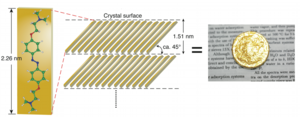
1) A. Matsumoto, M. Kawaharazuka, Y. Takahashi, N. Yoshino, T. Kawai, Y. Kondo*, J. Oleo Sci., 59(3), 151-156 (2010).
2) Y. Kondo*, A. Matsumoto, K. Fukuyasu, K. Nakajima, Y. Takahashi, Langmuir, 30(15) 4422-4426 (2014).
3) 橋 裕, 近藤行成*, 色材協会誌, 87(12), 442-447 (2014).
Evaluation and application of the structure and stability of foam formed by surfactants
We evaluate the structure and stability of foam formed by surfactants by combining various measurement techniques. In particular, we are working on establishing a "foam nano-operand measurement technique" using small-angle neutron scattering, which has few precedents, to reveal the ever-changing nanostructure of foam in a time-resolved manner. We also apply these foam properties to foam separation to recover hazardous substances and depleted resources.
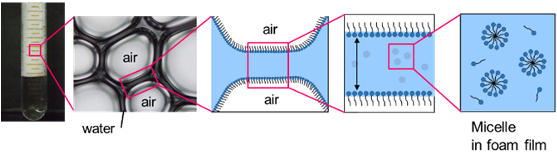
The microstructure of foam formed by hydroxy group-containing amino acid surfactants was clarified in detail by combining various measurement techniques such as small-angle neutron scattering

1) S. Yada*, H. Shimosegawa, H. Fujita, M. Yamada, Y. Matsue, T. Yoshimura*, Langmuir,36(27), 7808-7813 (2020). Selected as "Supplementary Cover"
2) 黒田瑞穂, 矢田詩歩, 吉村倫一, 大野正司, 好田年成, 多分岐鎖を有するポリオキシエチレン系非イオン界面活性剤のGaとAuの泡沫分離, 分離技術会年会 (2021).
3) 矢田詩歩, 永田夕佳, 吉村倫一, 中性子小角散乱を用いた単一鎖長ポリオキシエチレンアルキルエーテル硫酸エステルナトリウムによって安定化された泡沫のミクロ構造評価, 第73回コロイドおよび界面化学討論会 (2022).
Development of novel surfactants for high performance and high functionality
We have been developing surfactants with novel structures further to improve the performance and functionality of existing surfactants. For example, we have designed and synthesized a novel polyoxypropylene (PO)-EO surfactant with a PO chain modified at the end of the EO chain of a single-chain-length EO nonionic surfactant with no chain length distribution in the polyoxyethylene (EO) chain, and have demonstrated excellent surface tension lowering ability compared to existing EO surfactants. (PO-EO) chains. The aqueous solution properties of these surfactants are evaluated using cloud point, static and dynamic surface tension, as well as small angle X-ray scattering, small angle neutron scattering at synchrotron radiation facilities, dynamic light scattering, low-temperature transmission electron microscopy, and rheology.
1) S. Yada, T. Suzuki, S. Hashimoto, T. Yoshimura*, Langmuir, 33, 37394-3801 (2017).
2) S. Yada, T. Yoshimura*, Langmuir 35(15), 5241-5429 (2019).
3) S. Yada, M. Wakizaka, H. Shimosegawa, H. Fujita, M. Yamada, Y. Matsue, T. Yoshimura*,Colloids Surf. A, 611,125757 (2021).
4) S. Yada*, Y. Yoshioka, M. Ohno, T. Koda, T. Yoshimura*, Colloids Surf. A, 648,129247 (2022).